The Best MedTech Labeling Systems
Trusted by Smith and Nephew, Medtronic, Philips and many more.
We offer FDA-compliant labeling solutions to medical device firms, ensuring regulatory adherence and product safety through specialized labeling services.
Call Us: 408-748-9112

FDA/UDI Compliance
We help medical device and pharmaceutical businesses of all sizes meet compliance labeling regulations
We grow with you
Our systems are never one size fits all. We will advise and present options with your business needs in mind
PRINTERS, SUPPLIES AND SOFTWARE THAT ARE EASY TO USE.

Efficiency comes first
Our goal is to help you label your products in the way that fits your marketing, growth and compliance goals
we eliminate downtime
Planning alongside you we are able to make sure any label supplies you need are kept stocked and ready
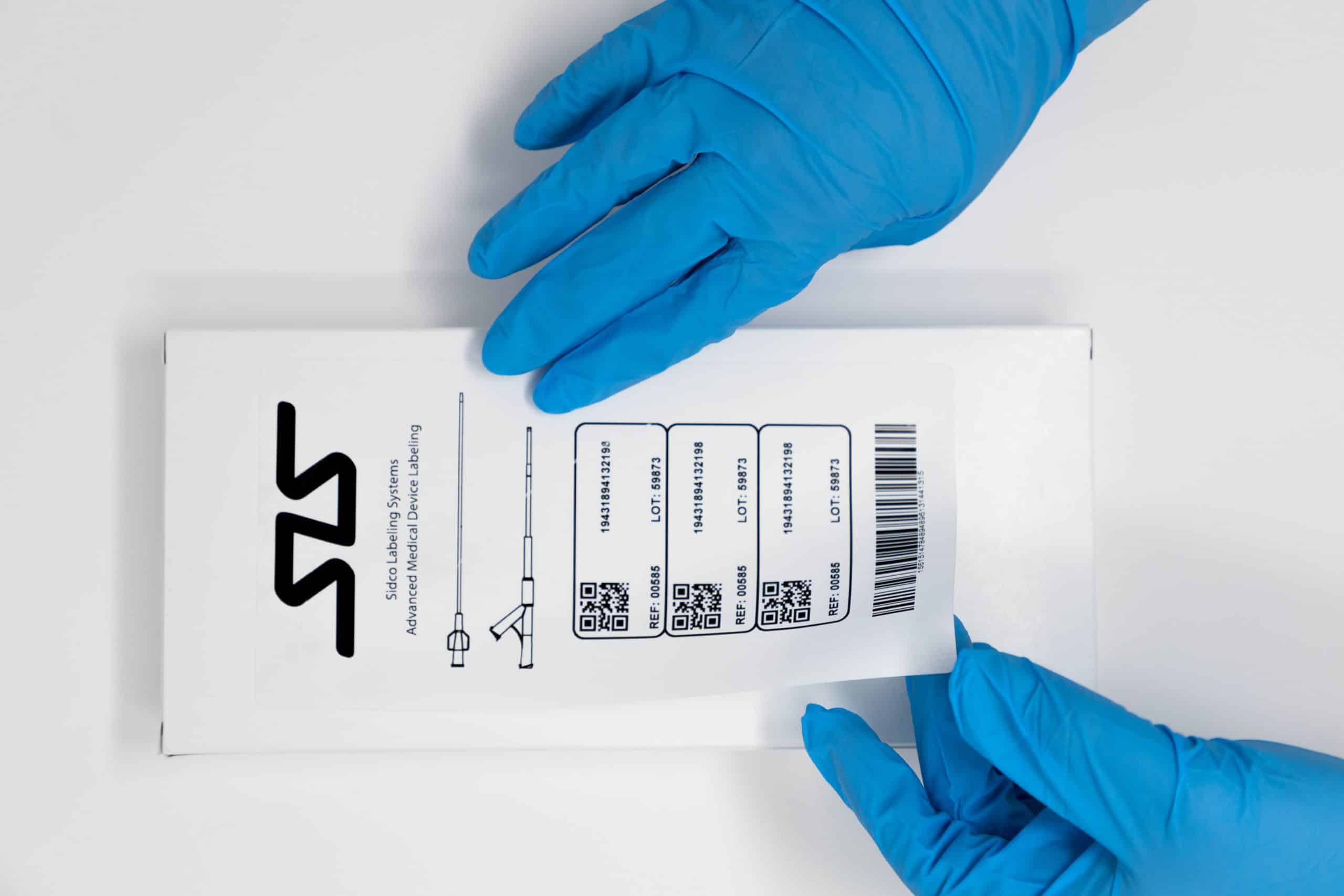
Who we are
Labeling experts with 30 years of experience.
With an impressive 30-year legacy in medical device labeling, our company boasts unparalleled expertise and knowledge, consistently exceeding industry standards and ensuring the utmost precision and compliance.
Medtech Labeling Services:
Everything you need to be compliant with FDA standards while maintaining efficiency and cost effectiveness.

Variable Data Printing
Lot numbers, expiration dates, unique device identifiers. We have everything you need to do it yourself, or we can do it for you!

Inventory Control
Software and label printing systems that let you maintain FDA compliant traceability of your inventory from receipt of goods to shipment.

Integrated Systems
All our hardware and software can seamlessly integrate into your existing IT infrastructure. We have the expertise to help you.
Testimonials
What Our Clients Say
Call Us: 408-748-9112
Schedule a consultation with an expert:
Let us know a date and time for a call, we would love to hear more about your Medtech labeling needs.
